Fabian Gerlinghaus & Omar Kurdi

Cellares is the first Integrated Development and Manufacturing Organization (IDMO), providing global cell therapy development and manufacturing services through an Industry 4.0 approach to the mass manufacture of the living drugs of the 21st century. The company enables biopharmaceutical partners to develop, scale, and commercialize cell therapies with the capacity, reliability, and economics required to meet total patient demand.
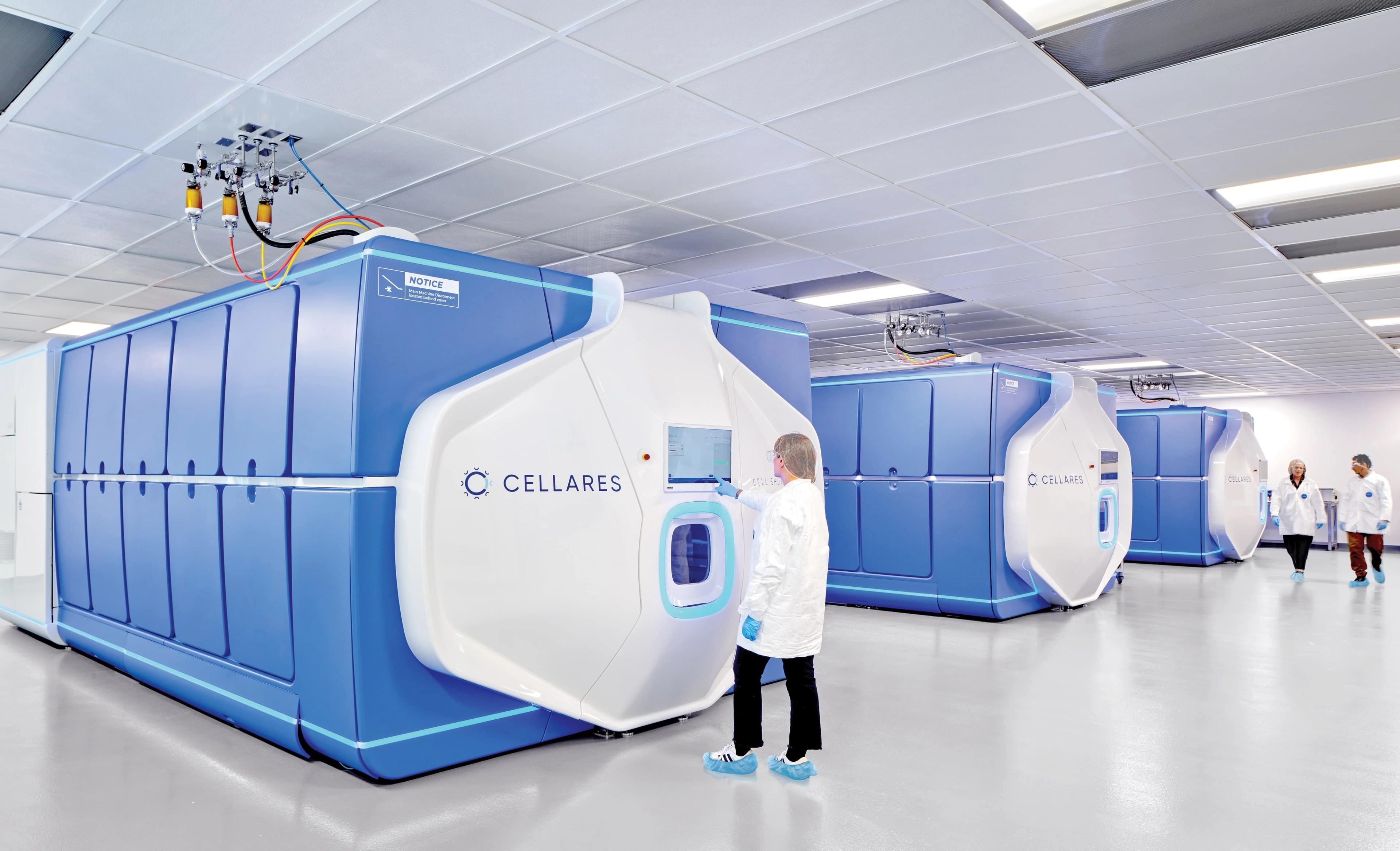
Cellares’ fully automated platforms — Cell Shuttle™ for end-to-end cell therapy manufacturing and Cell Q™ for automated in-process and release quality control — are deployed across its network of IDMO Smart Factories worldwide. These technologies deliver industry-leading manufacturing economics, higher process success rates, and the ability to produce up to 10× more cell therapy batches than conventional CDMOs with comparable footprint and headcount, resulting in the lowest cost of manufacturing in the industry.
Headquartered in South San Francisco, California, Cellares operates its first commercial-scale IDMO Smart Factory in Bridgewater, New Jersey, with additional facilities under construction in Europe and Japan. Through its global manufacturing network, Cellares is purpose-built to support both clinical and commercial programs and to expand access to life-saving cell therapies worldwide. For more information, visit www.cellares.com and follow Cellares on LinkedIn.
“We’re excited to accelerate access to personalized cures by providing therapy developers with cost-efficient and scalable manufacturing services.”
Co-Founder and CEO, Cellares
Fabian Gerlinghaus
Cellares Cell Shuttle™ for fully-automated and high-throughput cell therapy manufacturing.